Antibiotics in wastewater represent a growing and worrying menace for environmental and human health fostering the spread of antimicrobial resistance. Titanium dioxide (TiO2) is a well-studied and well-performing photocatalyst for wastewater treatment. However, it presents drawbacks linked with the high energy needed for its activation and the fast electron–hole pair recombination. In this work, TiO2 nanoparticles were decorated with Ag nanoparticles by a facile photochemical reduction method to obtain an increased photocatalytic response under visible light. Although similar materials have been reported, we advanced this field by performing a study of the photocatalytic mechanism for Ag–TiO2 nanoparticles (Ag–TiO2 NPs) under visible light taking in consideration also the rutile phase of the TiO2 nanoparticles. Moreover, we examined the Ag–TiO2 NPs photocatalytic performance against two antibiotics from the same family. The obtained Ag–TiO2 NPs were fully characterised. The results showed that Ag NPs (average size: 23.9 ± 18.3 nm) were homogeneously dispersed on the TiO2 surface and the photo-response of the Ag–TiO2 NPs was greatly enhanced in the visible light region when compared to TiO2 P25. Hence, the obtained Ag–TiO2 NPs showed excellent photocatalytic degradation efficiency towards the two fluoroquinolone-based antibiotics ciprofloxacin (92%) and norfloxacin (94%) after 240 min of visible light irradiation, demonstrating a possible application of these particles in wastewater treatment. In addition, it was also proved that, after five Ag–TiO2 NPs re-utilisations in consecutive ciprofloxacin photodegradation reactions, only a photocatalytic efficiency drop of 8% was observed. Scavengers experiments demonstrated that the photocatalytic mechanism of ciprofloxacin degradation in the presence of Ag–TiO2 NPs is mainly driven by holes and ˙OH radicals, and that the rutile phase in the system plays a crucial role. Finally, Ag–TiO2 NPs showed also antibacterial activity towards Escherichia coli (E. coli) opening the avenue for a possible use of this material in hospital wastewater treatment.
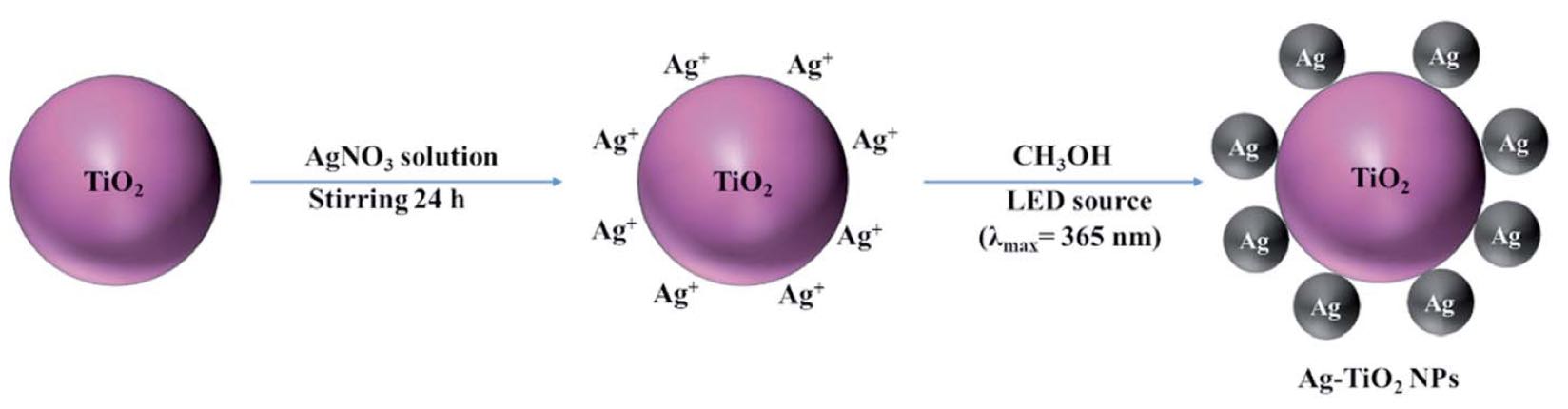
Antibiotics in wastewater represent a growing and worrying menace for environmental and human health fostering the spread of antimicrobial resistance. Titanium dioxide (TiO2) is a well-studied and well-performing photocatalyst for wastewater treatment. However, it presents drawbacks linked with the high energy needed for its activation and the fast electron–hole pair recombination. In this work, TiO2 nanoparticles were decorated with Ag nanoparticles by a facile photochemical reduction method to obtain an increased photocatalytic response under visible light. Although similar materials have been reported, we advanced this field by performing a study of the photocatalytic mechanism for Ag–TiO2 nanoparticles (Ag–TiO2 NPs) under visible light taking in consideration also the rutile phase of the TiO2 nanoparticles. Moreover, we examined the Ag–TiO2 NPs photocatalytic performance against two antibiotics from the same family. The obtained Ag–TiO2 NPs were fully characterised. The results showed that Ag NPs (average size: 23.9 ± 18.3 nm) were homogeneously dispersed on the TiO2 surface and the photo-response of the Ag–TiO2 NPs was greatly enhanced in the visible light region when compared to TiO2 P25. Hence, the obtained Ag–TiO2 NPs showed excellent photocatalytic degradation efficiency towards the two fluoroquinolone-based antibiotics ciprofloxacin (92%) and norfloxacin (94%) after 240 min of visible light irradiation, demonstrating a possible application of these particles in wastewater treatment. In addition, it was also proved that, after five Ag–TiO2 NPs re-utilisations in consecutive ciprofloxacin photodegradation reactions, only a photocatalytic efficiency drop of 8% was observed. Scavengers experiments demonstrated that the photocatalytic mechanism of ciprofloxacin degradation in the presence of Ag–TiO2 NPs is mainly driven by holes and ˙OH radicals, and that the rutile phase in the system plays a crucial role. Finally, Ag–TiO2 NPs showed also antibacterial activity towards Escherichia coli (E. coli) opening the avenue for a possible use of this material in hospital wastewater treatment.
